|
Problem 4: Calculate the pressure in pascals of 12,8 kilograms of methane (molar mass 16 g/mol) stored at 30 ☌ in the 70-liter storage tank of a methane-powered car. The volume V = 23.69 L at the normal conditions will be calculated.Enter the pressure and temperature for normal conditions P = 100 kPa, T = 0 ☌ and click or press the Calculate button.Change the Select an unknown selector to the Volume.The calculator will calculate the quantity in moles that will be used later.Calculate the volume of this mass of gas under normal conditions (P = 100 kPa, T = 0 °С). Problem 3: A pressure vessel is filled with a gas at a pressure P = 0.5 MPa and a temperature T = 15 ☌. Enter the volume V = 4 L and the pressure P = 3 MPa.The amount of oxygen in moles will be calculated.Enter the molar mass of oxygen M = 32 g/mol.Select the T (Temperature) in the Select an unknown selector.Solution: Click the Reset button and enter the problem data into the calculator: Determine the absolute temperature of 128 g of oxygen in a 4-liter vessel at a pressure of P = 3 MPa. Problem 2: The molar mass of oxygen gas (O₂) is M = 32 g/mol. The molar mass of the air will be calculated M = 0.029 kg/mole.Enter the mass m = 1.28 kg and click or press the Calculate button.The amount of 1 m 3 of the air in moles will be calculated. 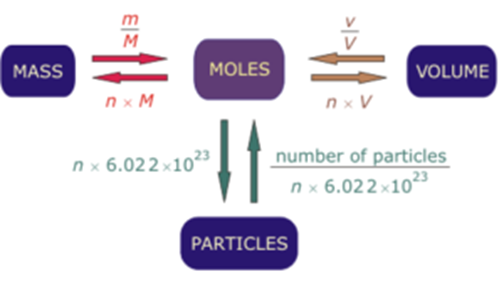
Enter the absolute pressure P = 100 kPa.Select the n (Amount in moles) in the Select an unknown selector.Click the Reset button and enter the problem data into the calculator: 
Solution: From the given air density we know that the mass of one cubic meter of air is 1.28 kg. Problem 1: Under normal conditions (temperature 0 ☌ and atmospheric absolute pressure 100 kPa), the air density is 1.28 kg/m³. Gay-Lussac’s Law ( V=const, n=const) Ideal Gas Practice Problems
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
